On September 28, 2021, the World Rabies Day, Trinomab announced that it will take full advantage of its core technology HitmAb® to develop a bivalent fully human anti-rabies neutralizing antibody cocktail, which is “highly effective, low toxicity and accessible”, indicating that the company has officially set foot in the rabies monoclonal drug field.
Rabies is a serious and lethal infectious disease, causing death close to 100%. Rabies causes more than 50,000 deaths worldwide every year, and the National Health Commission website shows 14 rabies cases and 8 deaths found in China in August 2021.

Figure 1. Statistics of reported morbidity and mortality of statutory infectious diseases nationwide in August 2021
The rabies vaccine cannot not block rabies virus completely but takes 7-14 days. For rabies post-exposure prophylaxis (PEP), rabies immunoglobulin (RIG) can rapidly trigger passive immune protection, but RIG is costly, of uncontrolled quality and carries the risk of transmission of other infectious diseases. In 2018, The World Health Organization (WHO) issued recommendations to use more standardized and quality-controlled anti-rabies virus monoclonal products as an alternative to RIG.
In July 2021, the FDA issued new guidance for the development of anti-rabies cocktails (Rabies: Developing Monoclonal Antibody Cocktails for the Passive Immunization Component of Post-Exposure Prophylaxis Guidance for Industry), specifying that monoclonal antibody drugs for post-exposure prophylaxis (PEP) consist of at least two rabies monoclonal antibodies in an antibody cocktail to respond to different rabies virus variants. In the future, the development of rabies cocktail antibodies will be an inevitable trend.

Figure 2. Rabies antibody development guidance issued by the FDA in July 2021
In China, about 15 million people are bitten by dogs and other animals each year, including about 7 million Class III exposures, resulting in an actual demand for rabies antibodies of 40 million doses, and compared to the annual batch issue of rabies immunoglobulin of 12 million doses (the stock market) there is still a potential incremental market of about 28 million doses. Therefore, there will be at least 8 billion yuan of market share for rabies antibodies when they go on sale (calculated at 200 yuan per dose for cocktail antibodies to rabies on sale). Together with India and Southeast Asia, the market for cocktail antibodies to rabies is expected to be at least 10 billion yuan globally.
Rabishield (monovalent human-derived monoclonal antibody) from Serum Institute of India and Twinrab (bivalent murine monoclonal antibody) from Zydus Cadila are currently available in India. Rabies monoclonal antibodies developed by North China Pharmaceutical Company are in the clinical stage.
The new guidelines bring new challenges and opportunities, and Trinomab will take full advantage of its core technology HitmAb® to develop a bivalent fully human anti-rabies neutralizing antibody cocktail, which is "highly effective, low toxicity and accessible", to set foot in the rabies monoclonal drug development field.
The antibody developed by Trinomab’s antibody discovery platform HitmAb® is a truly fully native human monoclonal antibody (Figure 3). Since it was was selected and matured in a human immune tolerance environment, the antibody can overcome the shortage of sources, unstable quality control, and the risk of infection with other infectious diseases that exist in rabies immunoglobulin, and will give patients the greatest assurance of effectiveness and safety in clinical practices.
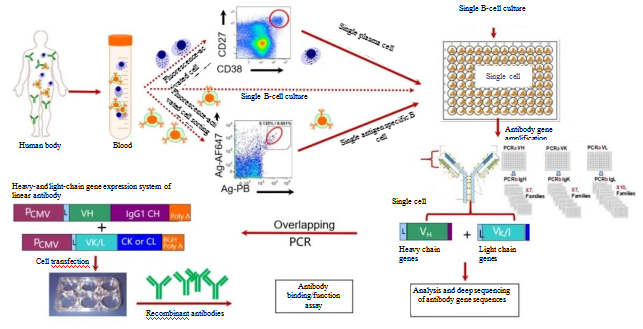
Figure 3. Trinomab’s HitmAb® technology roadmap
Trinomab has entered into a cooperation agreement with CMS (867.HK) to develop a bivalent fully human anti-rabies virus monoclonal antibody and a bivalent fully human anti-neocoronavirus monoclonal antibody in the form of a joint venture. This cooperation will fully integrate Trinomab’s efficient fully native human antibody discovery platform technology, molecular optimization and identification capabilities, its advantages in CMC, non-clinical and commercial production, as well as comprehensive strength in drug clinical development and marketing commercialization of CMS, to prevent rabies from killing patients in China and even around the world eventually by reinforcing complementary advantages.
About CMS
As a global innovation-driven and China-focused specialized pharmaceutical company, CMS (867.HK) is committed to providing competitive products and services to meet unmet medical, health and beauty needs in China. CMS focuses on innovative pipelines that are the first of their kind in the world, or have innovative formulations or delivery methods to achieve the best therapeutic effect in their class, or the safest, or the most beneficial ratio. The company now has more than 20 types of innovative products with high innovation grade, great market potential and competitive differentiation advantages. With the resources of experts and hospitals in various therapeutic areas, CMS is able to efficiently advance the clinical development of innovative drugs in China, while already creating leading market positions for many of its products on sale relying on strong commercialization capabilities. Driven by the combination of strong product power, excellent commercialization capability and efficient management system, CMS has been one of the most efficient pharmaceutical companies in China in terms of operational management.
About Us
As an innovative biopharmaceutical company at clinical stage with a perspective of global layout, Trinomab Biotech Co., Ltd (Trinomab for short) is mainly engaged in R&D of original fully native human mAbs new drugs. The core technology of the company is known as the fourth-generation antibody technology HitmAb, a proprietary technology platform featuring differentiated advantages and high efficiency for the discovery of fully native human mAbs against infectious diseases, autoimmune diseases (Patent: ZL201711013251.3), malignant tumors and other human diseases.
With the core technology of the “Integrated Technology Platform for the Development of Fully Native Human Monoclonal Antibodies (HitmAb®)” (Patent No.: ZL201711013251.3), Trinomab is committed to developing highly differentiated and highly effective fully native human monoclonal antibodies with independent intellectual property rights to enhance and improve the prevention and treatment of infectious diseases, autoimmune diseases, malignant tumors and other diseases.

Trinomab Biotech Co., Ltd.
Zhuhai R&D Center: 2F, Building 6C, Zhizao Avenue, Hongqi Town, Jinwan District, Zhuhai
Zhuhai Production Base: Building 9, International Health Port, Sanzao Town, Jinwan District, Zhuhai
Beijing Clinical Center: W2-911, Oriental Plaza, Dongcheng District, Beijing
U.S. Office: 4307 Forest Edge, Trail Durham, NC 27705. USA
Australia Office: 17 Bungowen Ave, Thornleigh, NSW Australia 2120
Tel.: 0756-7263999
E-mail: trinomab@trinomab.com